Neostigmine
Structure –
Neostigmine is a drug that is used to improve muscle strength and tone in conditions such as myasthenia gravis, a neuromuscular disorder. Its chemical structure is as follows:
The molecule has a carbamate group (-O-CO-NH-) attached to a quaternary nitrogen atom. It also has a methyl group (-CH3) attached to the carbamate nitrogen atom. This structure is important for the drug’s mechanism of action, which involves inhibiting the activity of the enzyme acetylcholinesterase, thereby increasing the levels of acetylcholine at neuromuscular junctions and improving muscle function.
Synthesis –
The synthesis of it involves several steps and is typically performed through a multi-step organic synthesis. Here is a brief overview of the process:
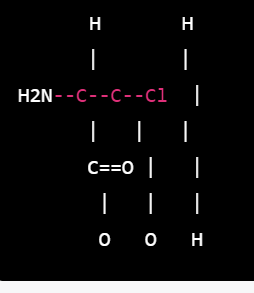
- The starting material for neostigmine synthesis is 3-dimethylaminophenol (also known as meta-aminophenol), which is reacted with chloroacetyl chloride to form a chloroacetylamino derivative.
- This chloroacetylamino derivative is then reacted with trimethylamine to form a quaternary ammonium salt.
- The quaternary ammonium salt is reacted with carbamate, which leads to the formation of neostigmine.
Overall, the synthesis of it is a complex process that requires careful control of reaction conditions and multiple purification steps to obtain a high-purity product. The synthetic route can be modified depending on the specific starting materials and reaction conditions used.
or
Here is the detailed synthesis of Neostigmine in structural formula:
Step 1: The starting material meta-aminophenol is reacted with chloroacetyl chloride in the presence of a base such as triethylamine to form a chloroacetylamino derivative.
Step 2: The chloroacetylamino derivative is reacted with trimethylamine to form a quaternary ammonium salt.
Step 3: The quaternary ammonium salt is reacted with carbamate, which leads to the formation of neostigmine.
This final compound is neostigmine, which is used as a medication to treat conditions such as myasthenia gravis.
SAR –
The SAR (Structure-Activity Relationship) of it refers to the relationship between the chemical structure of the drug and its pharmacological activity. Here are some key structural features of it that contribute to its activity:
- Quaternary ammonium group: The quaternary ammonium group in neostigmine is essential for its activity as an acetylcholinesterase inhibitor. This group interacts with the enzyme’s active site and prevents the breakdown of acetylcholine, thereby increasing its concentration at the neuromuscular junction.
- Carbamate group: The carbamate group in it is responsible for its high selectivity towards acetylcholinesterase. This group binds covalently to the enzyme, forming a stable complex that blocks its activity.
- Methyl group: The presence of a methyl group on the carbamate nitrogen atom of neostigmine contributes to its increased stability and selectivity towards acetylcholinesterase.
- Aromatic ring: The aromatic ring in neostigmine may play a role in enhancing its binding to the enzyme’s active site.
- Chain length: The chain length of the alkyl groups in the carbamate moiety also plays a role in the activity of neostigmine. Shorter chain lengths lead to increased potency, while longer chain lengths can decrease potency.
- Stereochemistry: The stereochemistry of neostigmine is important for its activity, as the (−)-enantiomer is more potent and selective than the (+)-enantiomer.
Overall, the SAR of neostigmine highlights the importance of the quaternary ammonium and carbamate groups in its activity as an acetylcholinesterase inhibitor, as well as the role of the methyl group, aromatic ring, chain length, and stereochemistry in optimizing its potency and selectivity.
Mechanism –
it is an acetylcholinesterase inhibitor that acts by increasing the levels of acetylcholine at the neuromuscular junction. Here is the mechanism of action of neostigmine:
- it enters the bloodstream and is distributed throughout the body, including to the neuromuscular junctions.
- At the neuromuscular junction, it binds to acetylcholinesterase, which is an enzyme that breaks down acetylcholine.
- By binding to acetylcholinesterase, neostigmine prevents the enzyme from breaking down acetylcholine, leading to an accumulation of acetylcholine in the synaptic cleft.
- The increased concentration of acetylcholine in the synaptic cleft leads to greater activation of nicotinic acetylcholine receptors on the postsynaptic membrane.
- Activation of the nicotinic acetylcholine receptors leads to an influx of sodium ions into the muscle fiber, which depolarizes the membrane and triggers muscle contraction.
- Neostigmine is eventually broken down by the body’s enzymes and eliminated from the system, which allows acetylcholinesterase to resume its normal function of breaking down acetylcholine.
Overall, it works by inhibiting the activity of acetylcholinesterase, leading to increased levels of acetylcholine and enhanced muscle function in conditions such as myasthenia gravis.
Uses –
Neostigmine is a medication used for several medical conditions related to muscle weakness, such as:
- Myasthenia gravis: it is commonly used to treat myasthenia gravis, a neuromuscular disorder characterized by muscle weakness and fatigue. Neostigmine works by increasing the levels of acetylcholine at the neuromuscular junction, which enhances muscle function.
- Postoperative urinary retention: it can be used to treat postoperative urinary retention, a condition where patients have difficulty urinating after surgery. Neostigmine works by stimulating the bladder muscles, which helps to improve urine flow.
- Reversal of neuromuscular blocking agents: it is also used to reverse the effects of neuromuscular blocking agents used during surgery. Neuromuscular blocking agents are drugs that temporarily paralyze muscles to facilitate surgical procedures, and neostigmine can be used to reverse this paralysis.
- Gastrointestinal motility disorders: it can be used to treat gastrointestinal motility disorders, such as gastroparesis, which is characterized by delayed gastric emptying. Neostigmine works by enhancing the activity of the muscles in the gastrointestinal tract, which improves motility.
Overall, it is a medication with a variety of uses in the treatment of muscle weakness and related conditions.